Table of Contents
- Overview
- Vaccination in Early Childhood
- Vaccination in Adolescence
- Vaccination During Pregnancy
- Vaccination in Older Adulthood
- Key Takeaways
- Additional Resources and References
Overview
Although children receive most immunizations during the first few years of life, when their immune systems are still developing and they are most vulnerable to disease, vaccines provide critical protection at every stage of life—from infancy through adolescence, pregnancy, and even old age. A life course approach to immunization focuses on optimizing vaccination for all ages, integrating immunization services with other age-specific interventions to maximize available resources and close immunity gaps that occur throughout a person’s life.
Vaccination in Early Childhood
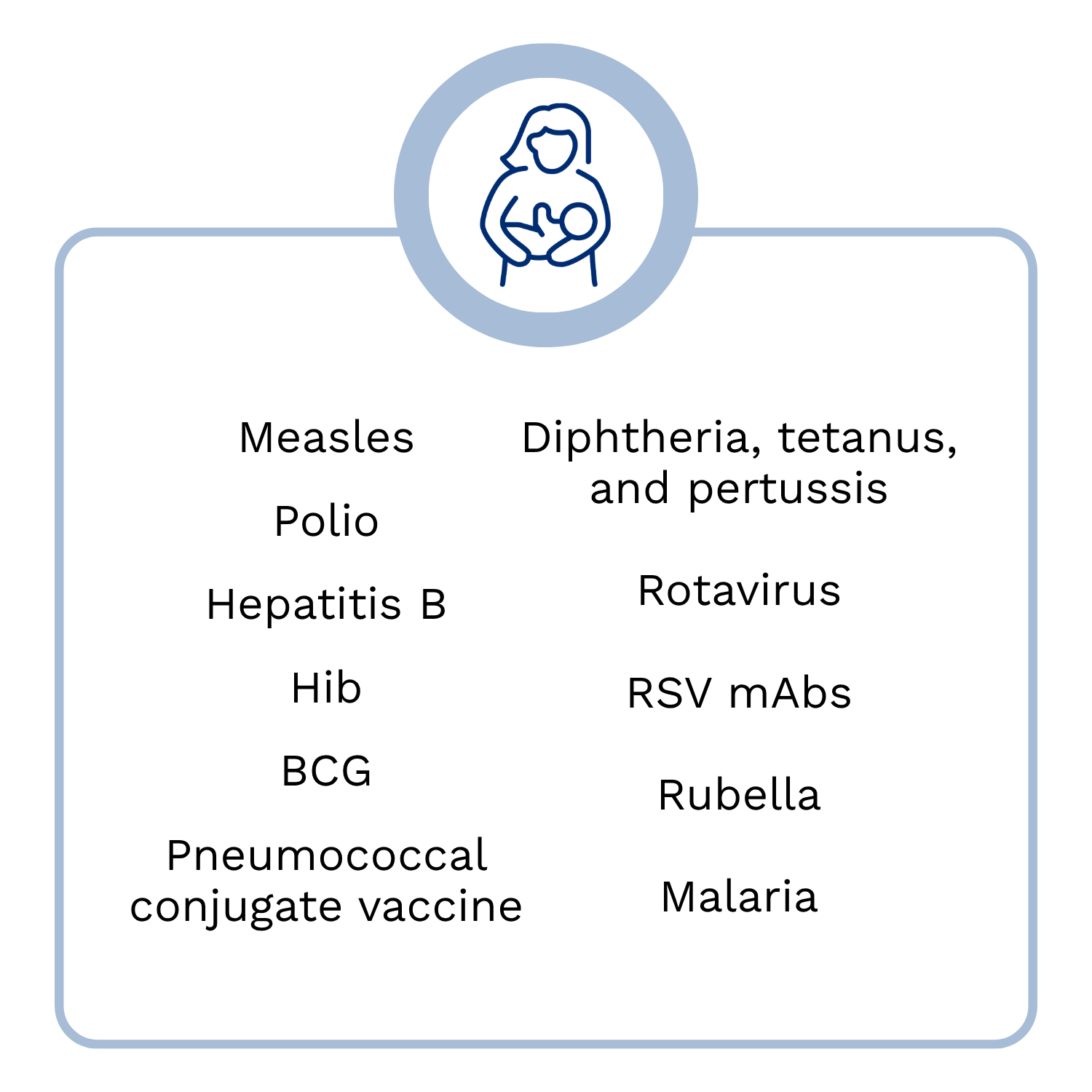
We receive more vaccines during infancy and early childhood than in any other stage of life to increase child survival, prevent disease outbreaks, and help children thrive. Over the past 50 years, immunization has had a significant impact on infant mortality and is estimated to have saved the lives of 101 million infants [1]. To continue these successes, it is critical that we maintain high coverage of longstanding childhood vaccines, facilitate uptake of those that have been more recently introduced, and close the remaining gaps in vaccine introduction. Further, although many countries have established effective programs to deliver vaccines to infants, strong platforms to reach children during their second year of life will need to be strengthened, as immunization for this age group is less likely to be prioritized by caregivers.
Measles
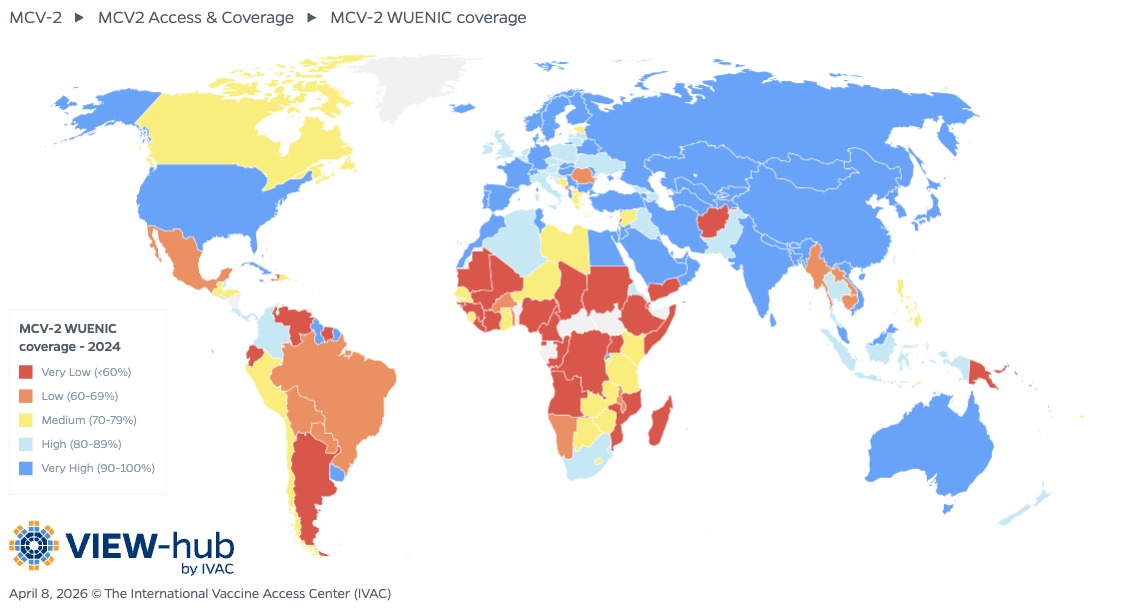
Although a single dose of measles-containing vaccine (MCV) can provide lifelong protection in most children, WHO recommends that all countries include a second dose (MCV2) in their national vaccination schedules, regardless of national MCV1 coverage, to ensure protection for the small percentage of children who do not respond to the first dose. MCV2 is most commonly administered during the second year of life and is thus a critical part of the second year of life platform for immunization. As of April 2026, all but two countries (Gabon and Central African Republic) have introduced two doses of MCV into their national immunization programs, although both countries are planning introduction of MCV2.
Many countries that were early to introduce MCV2 have been able to maintain or even increase coverage following COVID-19-related disruptions. For example, Zambia was able to increase national MCV2 coverage from 65% in 2018 to 77% in 2024. Further, several countries that have introduced MCV2 in the past few years have steadily increased coverage. Longstanding measles immunity gaps in the Democratic Republic of the Congo had previously contributed to increasing numbers of measles cases and frequent outbreaks, but after introducing MCV2 nationally in 2022, the country managed to achieve 51% coverage in 2024.
Rotavirus
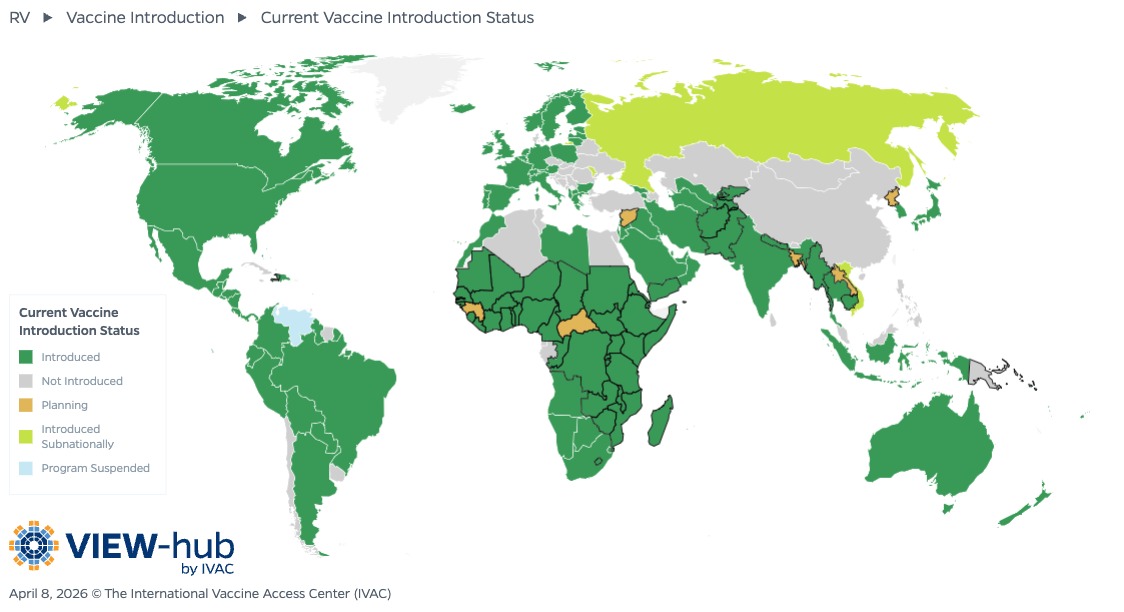
Over the past two decades, rotavirus vaccines have helped reduce the global burden of severe rotavirus gastroenteritis [2], which can lead to hospitalization or even death. As of April 2026, 140 countries have introduced rotavirus vaccines into their national immunization programs, including 46 Gavi-eligible countries. This number is up from 120 countries just five years ago, with recent introductions in Nigeria, Chad, South Sudan, and Cambodia, among other countries. Six additional Gavi-eligible countries are planning to introduce rotavirus vaccines into their national immunization programs, and three countries have introduced subnationally.
Of the countries that have not yet introduced rotavirus vaccination, several are in sub-Saharan Africa and South and South-eastern Asia, including Guinea, Central African Republic, Papua New Guinea, and Bangladesh, among others. As these regions are home to a large number of rotavirus deaths in children under 5 years [3], future efforts to advocate for rotavirus vaccine introduction should focus on these countries to have the biggest impact on child health.
PCV
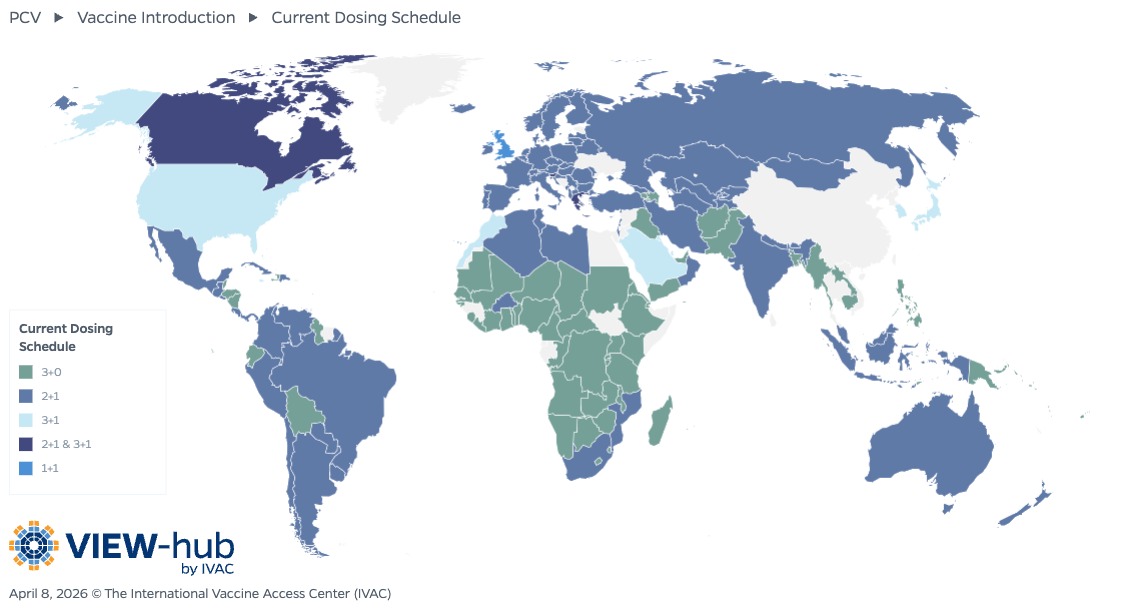
Pneumococcal conjugate vaccines (PCVs) have been highly effective at preventing childhood pneumococcal disease, an important cause of mortality among children under 5 years. As of April 2026, 177 (91%) countries have introduced PCVs, including 50 Gavi-eligible countries. An additional eight countries are planning introduction of PCVs into their national immunization programs. WHO recommends that countries achieve high coverage of three doses of PCVs by using either three primary doses without a booster dose (3+0) or two primary doses with one booster dose (2+1). Countries with mature PCV programs and adequate levels of population herd immunity can consider a reduced dosing schedule of one primary dose with one booster dose (1+1) to lower costs and give fewer injections, although this approach requires high vaccination coverage and close monitoring to ensure adequate protection.
Of countries that have introduced PCVs, about half of countries (85) use a 2+1 schedule, followed closely by those using a 3+0 schedule (69 countries). Just one country (United Kingdom) currently uses a 1+1 schedule. Maintaining high coverage of PCVs regardless of dosing schedule is essential for preventing childhood pneumonia deaths.
Vaccination in Adolescence
As children get older, there is generally less focus on their immunization needs and fewer touchpoints with the health system. However, vaccination remains a critical intervention among this population to combat the waning immunity to childhood vaccines as well as to provide protection against diseases that are unique to adolescents (such as invasive meningococcal disease, which adolescents are at increased risk for due to close contacts at school and other social behaviors) [4]. Human papillomavirus (HPV) vaccines are also recommended for this age group before they become sexually active to protect against cervical cancer and other cancers later in life.
HPV
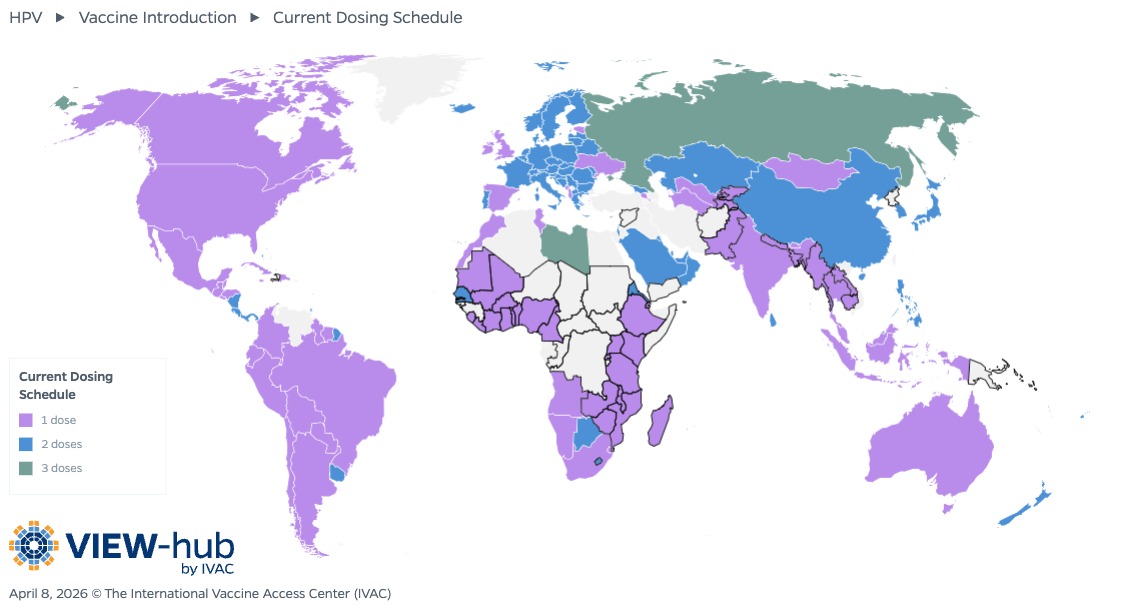
Although HPV vaccine introduction was originally limited to high-income countries, 161 countries of all income levels (including 36 Gavi-eligible countries) have introduced universal HPV vaccination as of April 2026, with another 19 countries planning introduction. These introductions have been made possible largely by the adoption of a single-dose schedule, which has been found to provide comparable protection to a multi-dose schedule while drastically reducing the cost per adolescent [5]. As of April 2026, more than half of countries (and 92% of Gavi-eligible countries) that include HPV vaccines in their national immunization programs have adopted a single-dose schedule. Of the 33 countries that have introduced HPV vaccination since the WHO included a single-dose schedule as an off-label alternative in December 2022, more than two-thirds (23) have introduced using a single-dose schedule.
Approximately 45% of countries that have introduced HPV vaccines use a two-dose schedule, and just two countries continue to use a three-dose schedule. As evidence continues to demonstrate the non-inferiority offered by a single dose of HPV vaccine [6], more countries are likely to adopt this strategy, allowing them to reach more adolescents with fewer resources.
Vaccination During Pregnancy

Pregnant women are at increased risk of severe disease and newborns’ immature immune systems leave them especially vulnerable to infectious diseases, making maternal immunization a key element of prenatal care to protect both mother and baby. Maternal vaccines have protected millions of newborns from influenza, pertussis, and tetanus, and newly licensed respiratory syncytial virus (RSV) vaccines can help to prevent severe disease in young infants [7-9].
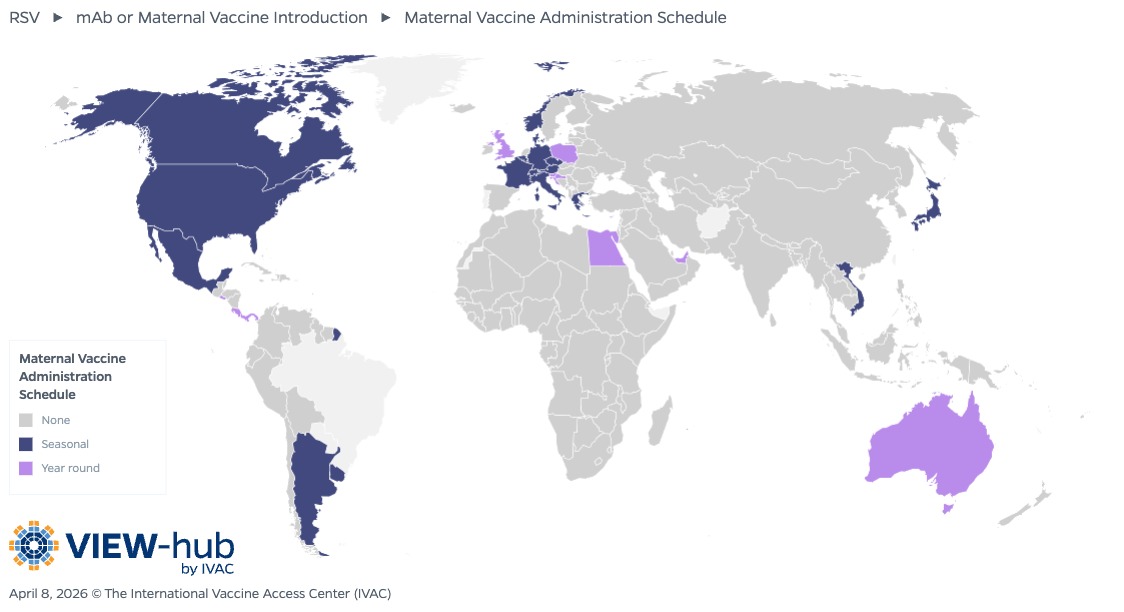
RSV
Prevention of RSV through maternal vaccines or infant monoclonal antibodies is critical, as there is no specific treatment for this potentially life-threatening respiratory virus. Maternal RSV vaccination provides passive immunization to infants by stimulating the production of antibodies to RSV in pregnant women, which are then transferred to their infants through the placenta. The first maternal vaccine (Abrysvo) became available in 2023 and was first introduced in countries in 2024. As of March 2026, 26 (13%) countries have introduced the RSV maternal vaccine. The countries that have introduced RSV maternal vaccination are primarily high-income countries (23), along with a few (3) upper middle-income countries. Notably, the vaccine has not yet been introduced in any low- or lower middle-income countries or Gavi-eligible countries, even though these countries carry a disproportionate burden of disease [10].
Coverage of maternal RSV vaccination varies, and efforts should focus on promoting uptake through targeted communication strategies to address vaccine hesitancy among this population. Further, additional advocacy is needed to ensure that pregnant women and infants everywhere have access to maternal RSV vaccination through continued introductions, particularly in those countries that would benefit most.
Vaccination in Older Adulthood

Older adults are more vulnerable to many infectious diseases due to age-related immune changes, resulting in increased risk of hospitalization for influenza, COVID-19, and RSV, among others [11-13]. The prevalence of comorbid conditions like chronic obstructive pulmonary disease and close living conditions in long-term care facilities also contribute to increased risk of pneumococcal disease, coupled with a higher rate of antibiotic resistance that can make treatment more challenging [14]. Despite this vulnerability, increasing vaccine access and uptake among older adults is an often overlooked approach to prevent disease and promote health, with many low- and lower-middle-income countries leaving adults out of national immunization strategies [15].
COVID-19 vaccination efforts demonstrated the potential to scale up adult immunization programs, with 84% of older adults in WHO member states completing the COVID-19 primary vaccination series by the end of 2023, although this success has not yet translated to other critical vaccines for older adults. In the United States, for example, uptake of RSV vaccination among adults aged 60 or older was just 16.4% during the first two seasons of vaccine availability, even among those at increased risk of severe illness due to existing health conditions [16].
Beyond reducing hospitalizations, vaccines can also have other benefits for this population. For example, shingles vaccination has been shown to have a protective effect against dementia diagnoses and can also slow the progression of disease among those already living with dementia [17,18]. As the older adult population is expected to continue growing, particularly in LMICs, efforts to increase vaccination among this age group should be prioritized.
Key Takeaways
- A life course approach to vaccination, which focuses on preventing disease from infancy through adulthood, is critical for closing immunity gaps and promoting lifelong health.
- The introduction of new childhood and adolescent vaccines such as those preventing rotavirus and HPV should be prioritized alongside efforts to increase coverage of vaccines that have already been implemented in national immunization programs.
- Maternal vaccination is key to protect pregnant women and their infants, though LMICs are currently left out of many of these efforts.
- Adult immunization programs must be strengthened to address the increased risk of vaccine-preventable diseases among a growing population of older adults.
References
- Shattock AJ, Johnson HC, Sim SY, Carter A, Lambach P, et al. Contribution of vaccination to improved survival and health: modelling 50 years of the Expanded Programme on Immunization. Lancet. 2024;403(10441):2307-2316. doi:10.1016/S0140-6736(24)00850-X
-
Aliabadi N, Antoni S, Mwenda JM, Weldegebriel G, Biey JNM, et al. Global impact of rotavirus vaccine introduction on rotavirus hospitalisations among children under 5 years of age, 2008-16: findings from the Global Rotavirus Surveillance Network. Lancet Glob Health. 2019;7(7):e893-e903. doi: 10.1016/S2214-109X(19)30207-4
-
Tate JE, Burton AH, Boschi-Pinto C, Parashar UD, Global Rotavirus Surveillance Network. Global, regional, and national estimates of rotavirus mortality in children <5 years of age, 2000–2013. Clin Infect Dis. 2016;62(Suppl 2):S96-S105. doi:10.1093/cid/civ1013
-
Burman C, Serra L, Nuttens C, Presa J, Palmer P, York L. Meningococcal disease in adolescents and young adults: a review of the rationale for prevention through vaccination. Hum Vaccin Immunother. 2018;15(2):459-469. doi:10.1080/21645515.2018.1528831
-
Slavkovsky R, Mvundura M, Debellut F, Naddumba T. Evaluating potential program cost savings with a single-dose HPV vaccination schedule: a modeling study. J Natl Cancer Inst Monogr. 2024;2024(67):371–378. doi:10.1093/jncimonographs/lgae037
-
PATH. New study validates previous conclusions supporting a single-dose HPV vaccination schedule. Gavi, the Vaccine Alliance VaccinesWork. Accessed March 27, 2026. https://www.gavi.org/vaccineswork/new-study-validates-previous-conclusions-supporting-single-dose-hpv-vaccination
-
Azziz-Baumgartner E, Grohskopf L, Patel M. Realizing the Potential of Maternal Influenza Vaccination. JAMA. 2021;325(22):2257–2259. doi:10.1001/jama.2021.7776
-
Briga M, Goult E, Brett TS, Rohani P, Domenech de Cellès M. Maternal pertussis immunization and the blunting of routine vaccine effectiveness: a meta-analysis and modeling study. Nat Commun. 2024;15:921. doi:10.1038/s41467-024-44943-7
-
Messeret ES, Masresha B, Yakubu A, Daniel F, Mihigo R, et al. Maternal and neonatal tetanus elimination (MNTE) in the WHO African region. J Immunol Sci. 2018;15:103-107. doi:10.29245/2578-3009/2018/si.1115
-
Lutz CS, Zhang H, Knoll MD, Sparrow EG, Chen H, Feikin DR. Respiratory syncytial virus positivity among hospital admissions for acute respiratory illness in children younger than 5 years of age in low- and middle-income countries: a systematic review and meta-analysis. BMC Public Health. 2026;26(1059). doi:10.1186/s12889-026-26743-4
-
Lafond KE, Porter RM, Whaley MJ, Suizan Z, Ran Z, et al. Global burden of influenza-associated lower respiratory tract infections and hospitalizations among adults: A systematic review and meta-analysis. PLoS Med. 2021;18(3):e1003550. doi:10.1371/journal.pmed.1003550
-
Taylor CA, Patel K, Pham H, Kirley PD, Kawasaki B, et al. COVID-19–Associated Hospitalizations Among U.S. Adults Aged ≥18 Years — COVID-NET, 12 States, October 2023–April 2024. MMWR Morb Mortal Wkly Rep. 2024;73(39):869–875. doi:10.15585/mmwr.mm7339a2
-
Burkart K, Liang C, Rafferty Q, Gillespie CW, McLaughlin S, et al. Respiratory syncytial virus-attributable hospitalizations among adults in high- and middle-income countries: application of the Global Burden of Disease framework. eClinicalMedicine. 2025;85(103292). doi:10.1016/j.eclinm.2025.103292
-
Cheong D, Song JY. Pneumococcal disease burden in high-risk older adults: Exploring impact of comorbidities, long-term care facilities, antibiotic resistance, and immunization policies through a narrative literature review. Hum Vaccin Immunother. 2024;20(1):2429235. doi:10.1080/21645515.2024.2429235
-
Vilajeliu A, Vega V, Gibson R, Nogareda F, Wang X, et al. Global Status of Adult Immunization Post COVID-19 Pandemic. Vaccines. 2025;11(13):4. doi:10.3390/vaccines13040401
-
La EM, McGuiness CB, Singer D, Yasuda M, Chen C. RSV vaccination uptake among adults aged 60 years and older in the United States during the 2023–2025 vaccination seasons. Hum Vaccin Immunother. 2025;21(1):2535755. doi:10.1080/21645515.2025.2535755
-
Eyting M, Xie M, Michalik F, Heß S, Chung S, Geldsetzer P. A natural experiment on the effect of herpes zoster vaccination on dementia. Nature. 2025;641(8062):438-446. doi:10.1038/s41586-025-08800-x
-
Xie M, Eyting M, Bommer C, Ahmed H, Geldsetzer P. The effect of shingles vaccination at different stages of the dementia disease course. Cell. 2025;188(25):7049-7064.e20. doi:10.1016/j.cell.2025.11.007